Formulation, Characterization, and Advantages of Using Liposomes in Multiple Therapies
Florina Miere (Groza)1*, Luminița Fritea2, Simona Cavalu2, Simona Ioana Vicaș3
|
|
|
Abstract
Liposomes or lipid nanoparticles are amphiphilic vesicles that allow the inclusion of substances of interest of any kind, both lipophilic and hydrophilic. This has put these transport systems in the spotlight for some time because it has been shown that they can function as carriers to release controlled and targeted substances of interest without being degraded. Thus, due to the major importance of these lipid vesicles in the development of the pharmaceutical industry and the medical field, in this paper we wanted to concentrate all information regarding physico-chemical properties, formulation methods, characterization techniques, stability and applications in various therapies and also highlighting some studies that have demonstrated the effectiveness of drug treatments of plant extracts transported by liposomes.
Keywords: liposomes, nanoparticles, characterization, applications
Introduction
Liposomes, as the name indicates, they are little spherical shaped artificial vesicles prepared by using various phospholipids [1-3]. Even if liposomes were first produced in 1961 by Alec D. Bangham, there is a continuous challenge for industrial and research domains such as biophysics, biochemistry, colloid science, and pharmacy, to find new fabrication methods and to extend their application area, especially towards advanced carriers and drug delivery tools and targeting specific tissue or organs [4, 5]. At the same time, optimization studies including prolonged half-life in blood circulation while avoiding harmful solvents used in the preparation route, are top research objectives.
Initially named “spherulites”, a mixture of lecithin and cholesterol dispersed in water was produced by handshaking or sonication, having a lamellar structure, as revealed by electron microscopy [6]. Nowadays, taking advantage of nano-technological tools, a particular kind of these particles have been touted as the next generation of liposomes, classified as nanoparticles [6]. These nanoparticles (or nanoliposomes) are lipids in nature because they are essentially made from a water emulsion in oil (W/O) or oil in water (O/W) [7, 8].
From a morphological point of view, liposomes are small (even nano-scaled) artificial vesicles, presenting one or more layers, able to incorporate a variety of both lipophilic and hydrophilic compounds [9, 10], and to release them at a targeted site in a suitable time.
Traditionally, the main constituents of liposomes are phospholipids: a hydrophilic “head” composed of three molecular components: choline, a phosphate group and glycerol, and two “tails” represented by a long chain containing essential fatty acids, with hydrophobic properties. The unique properties of such complex are auto-assembling and “self-sealing” in aqueous media, spontaneously entrapping bioactive compounds (Fig. 1) [11].
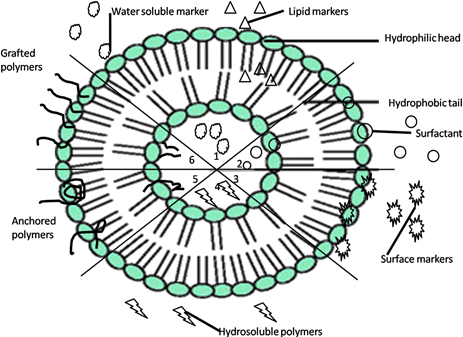
Fig.1.Schematic representation of unilamellar liposomes and positions of loaded substances (1-6). Adapted from [11]
Since now, a variety of fabrication methods were developed with the main goal to obtain different liposomal formulations. Unilamellar (UV) and multilamellar vesicles (MLV) were produced [7, 8], with the size ranging between 50-200nm, according to Harashima et al. [12]. Depending on the method used in the formulation process, the electric charge on the surface differs, and hence, Zeta potential is an important parameter which is responsible for the stability and encapsulation efficiency of the formulated vesicles [13-15], acting as a repulsive energy barrier and controlling the stability of dispersion and opposing the proximity of particles and aggregation. Knowledge of the zeta potential is also useful in controlling the aggregation, fusion, and precipitation of nanoliposomes, which are important factors affecting the stability of nano liposomal formulations [16].
This paper aims to briefly review the fabrication methods for the nano-liposomes formulation, their advantages, and disadvantages, as well as characterization methods at the laboratory level. Also, a review of different therapeutic active compounds encapsulated in nano-carriers targeting the desire organs or tissue is highlighted in the present review.
Advantages and Disadvantages of Lipid Nanoparticles over Conventional Transport Systems
The interest in the utilization of these liposomes in different pathologies is extremely high. The advantages and disadvantages of these structures can be traced in Table 1.
Table 1: Advantages and disadvantages of lipid nanoparticle transport systems [17, 18]
|
The advantages of liposomes |
The disadvantages of liposomes |
|
Biologically inert and biodegradable |
In case the support or matrix breaks or disintegrates too quickly, there is a risk of toxic effects, resulting in the release of a large dose of the drug at once |
|
Do not present the risk of toxicity, antigenicity or pyrogenicity because phospholipids are natural constituents of cell membranes |
The impossibility of abrupt discontinuation of treatment in the event of adverse reactions or the case of therapeutic accidents |
|
They can be prepared in different sizes, compositions, with different superficial tasks depending on the formulation requirements. |
The risk of accumulation if the rate of elimination of the drug substance is too slow |
|
They can encapsulate a wide variety of hydrophilic and lipophilic drug substances |
Poor absorption may delay the therapeutic action of the drug |
|
Protect the drug substances from the adverse action of the enzymes |
The period of the drug release is represented by the time the drug is passing through the gastrointestinal tract |
|
Rapid delivery and persistence of the active substance for a sufficient time at the plasma levels |
- |
|
Lower frequency of drug administration over a day |
- |
|
Decreased fluctuations in plasma levels |
- |
|
Reduction of the amount of active substance used |
- |
|
Release of the active substance at the target organ level, which leads to decreased frequency and intensity of adverse reactions and the use of lower doses |
- |
It can be observed that the advantages of these transport systems outweigh the disadvantages, which allows the use of these transport systems in fields such as medicine, pharmacy, biology, and technology because the risk is much lower than the benefits.
Physico-Chemical Properties of Liposomes
To obtain liposomes of nanometric dimensions, it is very important to know the physical and chemical properties of all the components that enter the structure of these vesicles. Therefore, great emphasis is placed on knowing the chemical composition of the substances used, and their transition temperature (Tc) [19].
The composition of liposomes is as follows:
• phospholipids - represent the major structural components of biological membranes;
• additives in the double phospholipid layers;
• cholesterol;
• enzyme activators, enzyme inhibitors;
• compounds for stabilizing liposomes;
• antioxidant agents;
• chelating agents.
There are two types of phospholipids, with their corresponding hydrolysis products: phosphoglycerides and sphingolipids.
Phospholipids are made up of glycerol, two fatty acids bound in positions l or 2 of glycerol, a phosphate group in position 3, and a polar group (Table 2) [20, 21].
Table2: Representation of the chemical structures of phospholipids
|
Abbreviation |
Name |
Structure |
|
PC |
Phosphatidyl-choline |
|
|
DLPC |
Diluaril-phosphatidylcholine |
|
|
DMPC |
Dimyristoyl-phosphatidylcholine |
|
|
DPPC |
Dipalmitoyl-phosphatidylcholine |
|
|
DSPC |
Distearoyl-phosphatidylcholine |
|
The most common phospholipids are phosphatidylcholines (PCs), also known as lecithins. They can be obtained from natural sources (eggs and soy) or by synthesis. These amphipathic molecules are hardly soluble in water. In the aqueous medium, they spontaneously align in double layers in the form of lamellar structures to reduce the unfavorable interactions between the aqueous phase and the long chains of fatty acids [22].
Lecithin from natural sources is a mixture of phosphatidylcholines, each with hydrocarbon chains of different sizes and different degrees of unsaturation. Plant lecithin has a high level of polyunsaturation, while mammalian sources contain a higher degree of fully saturated chains [23].
Sphingolipids are a versatile group of lipids that are made up of a sphinx (long alkyl chain gates) and a fatty acid that is linked by an amide group. They are present in the structure of eukaryotic cells with various roles such as activation or inhibition of protein kinases, signal transduction, or regulation of ion transport, the most important role being their involvement in cell apoptosis. As for their use, they can be used to formulate liposomes or to be incorporated into liposomes. Sphingolipids are most often embedded in liposomes, according to Kester et al., thus having a strong effect against cancer, the sphingolipid embedded and transported to tumor cells dictating cellular apoptosis. [24]
In the composition of the liposomal membrane, various compounds are also added, such as sphingomyelin which has the role of increasing the stability of immunoliposomes in Vivo, and the antigenic lipid cardiolipin used in immunological applications. Cholesterol is inserted into the liposomal membrane and due to the rigid sterol nucleus, it achieves a tight packaging of the lipid bilayer and improves the rigidity and hydrophobicity of the membrane. The ratio used by phospholipids: cholesterol must be 1:1 or 1:2 according to Mozafari et al. [7]. Phosphatidylcholine is the presence of cholesterol that increases liposomal permeability and may increase the fusion of liposomes with cells [25].
Searylamine, diethyl phosphate, and phosphatidic acid are used to stabilize liposomes. The addition of ionic compounds gives the liposomes a negative or positive charge, which causes an electrostatic repulsion between the different layers [26]. This results in an increase in interlamellar spaces with a higher encapsulation power for water-soluble substances [27]. EDTA is used as a chelating agent for metal ions that contaminate liposomes and can induce catalytic oxidation. Sodium chloride maintains isotonicity. Antifungal and antimicrobial agents improve the microbiological stability of liposomal preparations [28].
The transition temperature is defined as the temperature required to induce a change in the physical state of the lipids, from the gel phase, where the hydrocarbon chains are extended and closely linked, to the liquid-crystalline phase, where the hydrocarbon chains are randomly oriented. Several factors directly influence Tc, including the length of the hydrocarbon chain, the degree of unsaturation, the electrical charge.
It has been observed that Tc increases when the length of the hydrocarbon chain is longer, as the van der Waals interactions become stronger, requiring more energy to disorganize the membrane layer assembly. If a double bond is introduced into the acyl group, the energy of the unsaturated group is higher and thus a temperature lower than Tc is needed to produce the liposomal surface organization [29].
At the transition temperature, the two transition phases of liposomes coexist in equal proportions. At lower temperatures, the gel phase is reached and at higher temperatures, a disorganization of the fluid phase occurs [29].
Tc influences certain properties of liposomes such as permeability, aggregation, protein binding, and liposome fusion. The phase transition is used to induce the fusion of liposomes and the release of the active substance. Encapsulated substances can be released into the target tissue by modulating tissue temperature with various energy sources such as infrared radiation, microwaves, lasers. Tc is considered to be influenced by the nature of the phospholipids, the length of the acyl chain, the electrical charge, the degree of lipid saturation, and the nature of the existing groups. The shorter the acyl chain length, the lower Tc. It has also been shown that the release of doxorubicin for example is influenced by Tc [30]. There is a possibility of modifying or canceling the transition phase when serum or proteins adhere to the membrane surface.
Liposomes reach the transition state at the physiological temperature of 37 °C and can increase the phospholipase activity at the cell surface generating lysophospholipids (by deacylation in positions 1 and 2 to glycerol). These lysophospholipids administered to the spinal tissues of laboratory animals have been shown to produce neurotoxicity [31]. In addition to factors such as pH and electrical charge, the transition temperature greatly influences the process of obtaining liposomes. Tc is considered to be 10 °C higher than the melting temperature of the phospholipids used. It is called the transition temperature because at this temperature the permeability of the phospholipid membranes of the liposomes increases [7].
Methods of Formulation of Liposomal Systems
In the literature, there are a variety of methods that can be used to formulate liposomes or lipid nanoparticles [32]. They can be classified in different ways but depending on the type of nanoliposomes formulated there are the following categories which are shown in Fig.2.
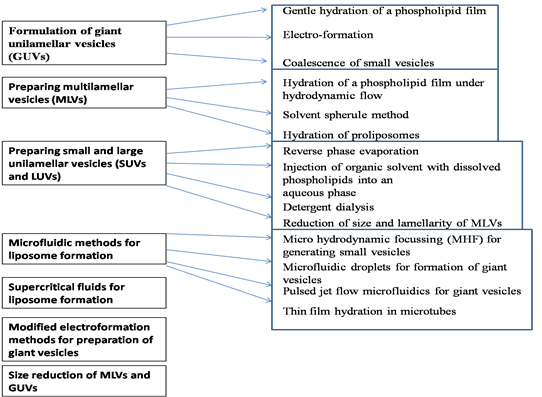
Fig. 2. Classification of nanoparticle formulation methods
The most important and most frequently used methods are sonication technique, extrusion method, and microfluidization. All these methods are applied after the formulation of the primary emulsion and have the role of shrinking the liposomes to nanometric dimensions, nanoliposomes, or nanoparticles [32].
Thus, the formation of the primary emulsion is carried out by the hydrated lipid film method, in other words, the phospholipids are mixed with different emulsifying agents (for example cholesterol) and solubilized in organic solvents (methanol, ethanol, chloroform). After solubilization, the lipid solution formed is subjected to evaporation using arotavapor to remove the solvent. Thus, on the walls of the balloon with a round bottom, the lipid film will form, which subsequently hydrates with aqueous solutions (distilled water, phosphate buffer solutions, HEPES buffer solutions). It is advisable to moisturize the buffer solutions which confers electrical charge on the surface of the liposomes formed at the contact of the two phases. This general formulation results in liposomes, large vesicles, and by the methods described later, the shrinking of the size is achieved, thus forming the nanoliposomes. A transition from the micrometric dimension of the liposomes to the nanometric dimension takes place, with the nano-type liposomes being considered vesicles with a size less than 100nm Fig.3.[33, 34].
Fig.3. Stages of the primary emulsion formulation
Characterization of Liposomes or Lipid Nanoparticles
To be able to demonstrate the success of liposome formation following the steps described above, a series of analyses are needed to prove the shape, size, electrical charge (Zeta potential) [35].
Optical and electronic methods can be used to visualize the shape of liposomes. For particles larger than 300 nm, the optical microscope can be used, but for particles smaller than 100 nm, the techniques of electron microscopy are most frequently used [35].
DLS (Dynamic light scattering): is used to determine the size of liposomes.
By injecting the samples in liquid form, graphs are obtained that highlight the size of the particles in suspension. This analysis can also determine the surface electrical charge of the liposomes, an important feature for maintaining liposome stabilization. This electric charge determines the Zeta-potential. Depending on the composition of the phospholipid "head" and the pH of the medium, liposomes can be charged with either a negative, positive, or neutral charge. The nature and density of the electric charge on the surface of the liposomal membrane influence the stability, the kinetics of biodistribution. as well as the uptake of liposomes by target cells[36].
Neutral liposomes have a lower tendency to be recognized and then destroyed by cells of the reticuloendothelial system after systemic administration. The main drawback of this type of liposome is that it has a greater tendency to form aggregates [37].
Negative liposomes: form stable suspensions and have low aggregation capacity [37].
Those containing phosphatidylserine (PS) or phosphatidylglycerol (PG) have a higher endocytosis rate because the negative surface is recognized by macrophage receptors.
The addition of glycolipids such as gangliosides or phosphatidylinositol inhibits the uptake of liposomes by macrophages and cells of the reticuloendothelial system, prolonging the circulation time. A small amount of negatively charged lipids stabilizes neutral liposomes against the aggregation mechanism [38].
Positive liposomes have a high tendency to interact with serum proteins and are easily recognized by macrophages with high hepatic, splenic, and pulmonary clearance. They are used in gene therapy for the delivery of intracellular DNA [38].
Microscopic techniques used to characterize the shape of liposomes
Microscopic techniques allow the visualization of the shape of liposomes and their morphology. Regarding the microscopic methods, they can be of several types: optical microscopy or electron microscopy [39]. In the following, they will be described in turns because each microscopic technique has both advantages and disadvantages [40].
Optical microscopy uses visible light, being easier to manipulate, but does not provide detailed results on the liposomes studied [41]. This microscopy does not allow the visualization of the lipid bilayer of the liposomes but allows the visualization of their shape. Therefore, this technique is slightly limited, but also, other types of optical microscopies can be performed, such as polarization, fluorescence, or confocal microscopy[42].
Polarization microscopy is slightly more advanced than conventional microscopy, allowing easy evaluation of the shape of liposomes, but like classical microscopy, it cannot provide details on the lipid bilayer of nanoscale liposomes. It can still provide a monochrome or colored image with a good resolution if the liposomes are micrometric in size [43].
A more advanced technique is fluorescence microscopy. This type of microscopy uses fluorochromes that can emit fluorescent light following light excitation at a certain wavelength [44]. The method is much more complex than described above and allows a more detailed view of the liposomes [44]. For example, in addition to their shape, the lipidic layers of the liposomal membrane can be visualized using rhodamine as a fluorescent substance. However, it also has certain disadvantages such as the possibility of destroying the liposomal membrane by injecting contrast agents (in quantities greater than 1%) or discoloration, whitening, and loss of fluorescence by prolonged exposure to the fluorescent light of the injected substance. Confocal microscopy is a more advanced version of fluorescence microscopy, and it can be used to obtain three-dimensional images of liposomes [45].
Unlike optical microscopy, electron microscopy allows the visualization of very small, nanometric liposomes. Electron microscopy uses electrons to obtain a projected image and includes scanning electron microscopy (SEM), transmission electron microscopy (TEM) [46].
SEM uses an electron beam that will result in an enlarged liposome image. Thus, much more complex information can be obtained on the shape, morphology, and structure of liposomes. However, because liposomes are quite difficult to apply and dry on the microscope support and because the vacuum created can deform the liposomal membrane, this method is quite rarely used [47].
TEM is the most common method used to characterize nanometric-sized liposomes. This method allows the use of an emulsion of nanoparticles in liquid form and the characterization of nanostructures with a size of 1nm [48].
In addition to these types of microscopy, optics and electronics can also be used for nanoparticle visualization and atomic force microscopy (AFM)[48].
AFM has an exceptional resolution, providing clear, three-dimensional images, concerning changes in the membrane of nanoparticles (attachment of antibodies for example), shape, morphology and can also provide mechanical and chemical information of the membrane structure [49].
Therefore, the choice of the liposome visualization technique is made taking into account the advantages and disadvantages of each method.
Liposome Stability
The shelf life of a lipid-containing product can be dramatically affected by the lipid species used in the formulation. Usually, the more double bonds the lipids contain in their structure, the easier they are to be degraded by oxidation. Lipids from biological sources contain large amounts of polyunsaturated fatty acids and hence their low oxidation stability, unlike usually saturated synthetic lipids [50].
Saturated lipids provide the highest stability against oxidation but have the disadvantage of a high transition temperature which makes them difficult to use in formulations. Another issue related to stability is hydrolytic degradation. Hydrolysis depends on many factors: pH, temperature, type of buffer, ionic strength, length of fatty acid chains, state of aggregation, and the possibility of water penetration into the membrane [50].
Among the stabilization methods, the following are known: the addition of antioxidants such as α-tocopherol, the addition of carbohydrates that are intercalated in the membrane, and can displace water from the region, drying lipids [29, 50]. Over time, changes in the structure of liposomes can occur due to the appearance of chemical degradation (oxidation and hydrolysis) but also physical degradation (aggregation, fusion, loss of the incorporated substance [51].
Prevention of chemical degradation
The level of oxidation can be reduced if precautions are taken including the use of freshly purified lipids and freshly distilled solvents, avoiding the use of processes that require high temperatures. It also takes into account the execution of processes in the absence of oxygen and deoxygenation of aqueous solutions with nitrogen. The storage of liposomal suspensions must be done in an inert atmosphere. It is also mandatory to include in the lipid membrane an antioxidant compound such as α-tocopherol, an iron chelator to prevent the initiation of a radical chain reaction [51]. The degree of saturation of the lipids used is important, being desirable to use saturated lipids instead of unsaturated ones susceptible to degradation due to oxidation. The pH used must be close to neutral [52].
Prevention of physical degradation
By physical degradation in the case of liposomes can be understood in terms of aggregation, sedimentation, fusion, which occur over a long time [52].
Aggregation and sedimentation of neutral liposomes occur due to van der Waals interactions and tend to increase in large liposomes where there are large membranous surfaces that allow contact between them. To avoid this phenomenon, a small amount of negative charge of phosphatidylglycerol or phosphatidic acid is introduced into the medium [53]. The reduction of the conversion of small liposomes into large liposomes due to fusion occurs by the addition of traces of the 1,3-diacyl-2-phosphatidylcholine isomer. The fusion of small liposomes can be prevented if they are stored at lower Tc temperatures and if enough cholesterol is included in their structure [54].
The permeability of the liposomal membrane depends on the composition of the lipid membrane and the entrained solute. Large polar molecules or ions will be easier to trap than lightweight lipophilic compounds. In general, for both types of compounds, the use of a rigid membrane saturated with a high percentage of cholesterol will lead to a reduction in the loss of entrapment [55].
Other methods of increasing membrane strength are fixation of glutaraldehyde in the membrane, osmification, inclusion of alkyl polymers such as polyvinyl alcohol esterified with stearic or palmitic acid, the inclusion of sphingomyelin or 2-carbamoylphosphatidylcholine derivatives [56].
The best solution for maintaining liposome stability is to transform liposomes into a solid, anhydrous form that decreases the possibility of degradation [52].
Liposomes Applications in Different Therapies
Over time, several issues have been addressed concerning the inclusion of substances or mixtures of substances in these transport systems. Both drugs such as acyclovir, betamethasone, retinol, atorvastatin, insulin, doxorubicin, etc. were incorporated, as well as plant extracts.
To date, numerous studies are testing in vitro the use of liposomes with trapped substances of interest. Liposomes do not have independent therapeutic activity and are used only as transport vectors of the included substance; therefore, their therapeutic activity is dictated by the incorporated substance. Thus, some liposomes have been tested in vitro in multiple diseases: heart therapy, skin diseases, respiratory diseases, eye diseases, in the fight against obesity, in the therapy of various types of cancer, liver diseases, etc.
Skin applications:the use of lipid nanoparticles in skin diseases is a topic of major interest because this innovative method allows the application of various substances with therapeutic action to the skin [57]. Nanoparticles can be used in the field of dermatology either for therapeutic purposes or for cosmetic purposes (Nanobase® or Cutanova® bases for example) [58]. The use of lipid nanoparticles in dermatological therapies has major advantages: due to their lipid-rich structure, they allow deep penetration of the dermis and retention of therapeutic substances, so a smaller amount of substance is used and the effect is more intense. These nano-sized particles will also keep the skin hydrated due to the occlusive film created on the surface of the skin [59].
Lipid nanoparticles are also used for the treatment of alopecia because they can penetrate through the follicular areas due to their nano-size. Research has also been done on the idea of combining nanoemulsions with moisturizers, this idea being the most promising on the skin and at the follicular level [60].
So far, they have been incorporated into lipid nanoparticles for cutaneous applications of vitamin A by Jeon et al., the effectiveness at the skin level being almost 5 times higher than the application of the vitamin as such. The same researchers have demonstrated in vivo in mice that nanoparticles with vitamin A have a major role in hair growth or that they can be used for anti-aging purposes by being incorporated into various creams [61].
Photoprotective substances have also been incorporated into the nanostructures and have been shown to have increased efficiency in this form. Puglia et al., [62].
Yue et al., 2010, Schwarz et al., 2013 incorporated and demonstrated that Coenzyme Q10 has a much higher efficiency in this form for sun protection and as antioxidant activity [63]. Lycopene also has an increased antioxidant and photoprotective effect if incorporated into nanoparticles according to Okonogi et al. [64]. Flavonoids have also been investigated, concluding that the cutaneous anti-inflammatory effect of quercetin is increased by incorporation into these therapeutic systems[65].
In some articles, the in vitro activity of Callendula Officinalis extract was highlighted by measuring the degree of healing of artificially obtained wounds with 48 hours [66].
Thus Lide Arana, Salado, et al., artificially created epithelial cell cultures that they "scratched" with a 0.22 μm pipette, then over these "wounds" they added the marigold extract as such and the marigold extracts embedded in the lipid nanoparticles. It was subsequently followed for 48 hours to assess the degree of epithelialization. Epithelial cells to which marigold extract was added showed a reepithelialization of the wound in a percentage of 20% at concentrations of 5g / ml after 48h. In the case of epithelial cells with marigold extract incorporated in lipid nanoparticles, a reepithelialization is observed in a higher percentage of 64.6% at concentrations of 5, 10, 20 µg / ml [66].
Medical substances such as minoxidil were also encapsulated for hair growth, fluocinolone acetate was incorporated for anti-inflammatory purposes, psoriasis, and ichthyosis were incorporated tretinoin, for the treatment of acne, hydroquinone was used for hyperpigmentation. All these substances also have an increased activity compared to the application of the substance as such [67, 68].
Vaccine carriers:in recent years, vaccine technology has been extensively studied for the inclusion of viral strains within liposomes. Thus, DNA, RNA, or various viral peptides reaching sizes up to 150nmcan be included in liposomes. This makes the intramuscular administration of the vaccine possible by including in liposomes the protection of these embedded compounds. The vaccine is thus more efficient, the liposome itself being an immune adjuvant factor in itself [69].
Heart disease:D. moldavica is a plant that belongs to the Lamiaceae family. The pharmacological effects of the extract of this plant are multiple, but the most important one is the effect exerted on the myocardium, thus acting in coronary heart disease, atherosclerosis, hypertension. All these actions are due to the flavonoid content. The aim was to include the extract in lipid nanoparticles because the bioavailability and solubility of the active ingredients are much lower if administered in a conventional pharmaceutical form. Thus, the administration of these nanoparticles containing flavonoids can be administered orally to combat cardiac ischemia [70]. The same authors evaluated the therapeutic capacity of flavonoid-loaded lipid nanoparticles from D. moldavica extract in myocardial diseases, such as cardiac ischemia. Thus, the authors obtained in vitro release results of the active principles from nanoparticles in a percentage of 96.23% while the release of the active substances from the emulsion itself was lower, 86.51% [70].
Pulmonary disease:Yun Zhao Yue-Xing et.al demonstrated in [71] that Yuxingcao extract encapsulated in solid nanoparticles has a higher pulmonary recurrence in vivo and therefore presents more prompt effect compared to the administration of the extract as such. Thus, the administration by nebulization of solid lipid nanoparticles loaded with Yuxingcao extract has a more intense and longer-lasting bronchodilator effect and can be used in various respiratory diseases [71].
Cancer therapy: doxorubicin is an antineoplastic active agent used to treat many types of cancer, namely lymphomas, leukemias, and solid tumors. However, its administration causes bone marrow depression, alopecia, and oral ulcers. The entrapment of DXR in liposomes decreases the toxicity of the substance and maintains or even increases the anti-cancer effect. Doxorubicin hydrochloride (DOXIL) is the first licensed liposomal product in the United States [72].
Research has been done to improve the safety profile of cytotoxic anthracyclines, doxorubicin (DXR) and daunorubicin (DNR), together with vincristine (VCR), which are associated with severe cardiotoxic and gastrointestinal side effects. The entrapment of these substances has led to a reduction in cardiotoxicity and dermal toxicity [72].
Cisplatin is another liposome-containing anticancer substance whose bioavailability is increased due to this fact. The advantage of including anticancer substances in liposomes is that the substances are "concentrated" in the tumor with a more prompt effect [73].
Letícia Mazzarino et al. demonstrated in [74] the antioxidant character of Jaboticaba extract by the DPPH and the FRAP method. Both methods were applied comparatively between the emulsion containing Jaboticaba extract and the extract as such. The results obtained were the following: both in the case of the DPPH method and the case of the FRAP method, it was shown that the extract incorporated in the nanoparticles has a more intense antioxidant activity, unlike the extract used as such, and it can be used in cancer therapy [74].
Eye disease: various anti-inflammatory substances that have been included in liposomal systems have also been employed for the treatment of the inflammation of the precorneal area and the cornea. These include diclofenac, ibuprofen, flurbiprofen, indomethacin, triamcinolone, all of which have better bioavailability and fewer liposomal side effects. In glaucoma, liposomal systems with timolol have been studied [75].
Another disease: In vitro entrapment of neomycin and penicillin into liposomes is effective in contrast to the antibacterial activity of chloramphenicol, which decreases greatly when embedded in these vesicles. Intravenous administration to mice in liposome-encapsulated modified gentamicin alters its tissue distribution, but it has been shown that intramuscular administration leads to sustained release at the injection site with prolonged plasma concentration [76].
Trapping of rifabutin in liposomes leads to a significant increase in activity against Mycobacterium avium and Mycobacterium tuberculosis infections compared to free rifabutin. The same can be seen with the anti-tuberculosis chemotherapeutic rifampicin, whose activity was increased dramatically when encapsulated in phosphatidylcholine liposomes. An increase in antituberculosis activity is observed when the tuftsin tetrapeptide macrophage activator is attached to the liposome surface [77].
Encapsulated clarithromycin is more effective against in vivo intracellular Mycobacterium avium infections, and ethambutol therapy may further increase the efficacy of the treatment [78].
The current treatment of immunocompromised patients infected with Mycobacterium avium is ineffective, probably because HIV is found to be inoculated intracellularly into monocytes. Effective treatment for the eradication of Mycobacterium avium complex disease in mice was obtained using amikacin. Amikacin incorporated into liposomes has also been shown to be useful in the treatment of extracellular infections with Klebsiella pneumoniae and Pseudomonas endocarditis due to its small, long half-life, and its ability to accumulate tissue [78].
Conclusions
This review focused on the formulation, characterization, storage, and use of liposomal forms. It can be concluded that liposomes or lipid nanoparticles are vesicular forms that do not have independent activity but can react differently depending on the environment and incorporated drugs or extracts with both hydrophilic and lipophilic behavior. Thus, these transport systems will protect the incorporated active principle and will convey it in a controlled and targeted way. This paper also highlights the chemical composition of liposomes and their widespread use in various types of diseases.
Therefore, these liposomal forms represent the future design of drugs or food supplements, the incorporated substance having a much greater efficacy, demonstrated in vitro, than the same substance found in the form of classical therapeutic models.
Acknowledgments
The author Miere (Groza) Florina thanks University of Oradea, Doctoral School of Biomedical Science, for providing assistance.
This work was funded by the Project 123008, "SmartDoct - High quality programs for doctoral students and postdoctoral researchers of the University of Oradea for the increase of the relevance of research and innovation in the context of the regional economy", project funded by the Operational Programme Human Capital 2014-2020.
References
