Enhancement of Solubility and Dissolution of Clopidogrel Bisulfate by Solid Dispersion in Combination with Surface Adsorbent
Priyal Patel*, Ashok Mahajan, NileeThakor
|
|
Department of Pharmaceutics, Babaria Institute of Pharmacy, BITS Edu Campus, Vadodara. Gujarat. India. |
ABSTRACT
The main objective of this work was to prepare solid dispersion adsorbate to improve solubility, dissolution of poorly water-soluble drug Clopidogrelbisulfate. The various carriers like PEG 4000, PEG 6000, Poloxomer 188, and Locust bean gum were used to prepare solid dispersion of Clopidogrelbisulfate, having different ratios using two methods solvent evaporation and hot melt method. Clopidogrelbisulfate containing surface solid dispersion was prepared using different carriers in different ratios and Neusilin as adsorbent by using two solvent evaporation and hot melt methods. To determine the effect of formulation variables, 32 Factorial design was applied. The angle of repose, % DE30 and Q30 were selected as dependent variables. Using Design Expert software (version 7.0.0) the optimized batch of Clopidogrelbisulfate was derived statistically. The animal study of the optimized batch was performed using the Albino rat where bleeding time and blood volume were checked. In solid dispersion, PEG 4000 gave the highest amount of drug release (1:2 ratio of drug:polymer) compared to other carriers when prepared by the solvent evaporation method. The tablets containing Clopidogrelbisulfate, PEG 4000, and Neusilin in a ratio of 1:2:1 were considered as optimized formulation. The Clopidogrelbisulfate containing an optimized formulation of surface solid dispersion showed 85.69% drug release in 30 min. The conversion from crystalline to the amorphous form of Clopidogrelbisulfate was confirmed by XRD studies. From the platelet aggregation inhibition study, it was concluded that when the rat was treated with surface solid dispersion, the bleeding time and the collected blood volume increased.
Keywords: Surface solid dispersion, Neusilin, Gibbs free energy, Platelet aggregation inhibition, Phase solubility, x-ray diffraction
Introduction
The Solid Dispersion technique is the most promising technique for improving the solubility and bioavailability of poorly water-soluble drugs. The hydrophobic drug is dispersed into a hydrophilic inert carrier using different techniques. The most commonly used carriers are PVP, Tween 80, Gelucire, PEG, etc. There are different methods of solid dispersion like spray drying, SCF technology, fusion method, lyophilization method, electrospinning method, and solvent method. However, some disadvantages are associated with the techniques that limit its use in the pharmaceutical industry like stickiness in the product, poor flow property, flow compressibility, less stability, and poor wettability. In order to overcome this, solid dispersion adsorbate is nowadays popular in the pharmaceutical industry. [1, 2] The Surface Solid Dispersion technique is defined as dispersing one or more active ingredients on a water-insoluble carrier of extremely high surface area to achieve increased bioavailability. The carrier used in surface solid dispersion is a porous material, which is hydrophilic in nature. These provide a larger surface area to the drug particles. The drug release from the carrier depends on porosity, particle size, and surface area of the carrier. In the solid dispersion method, the conversion in amorphous to the crystalline form of drug particles occurs, which is prevented by using adsorbent. Adsorbent having inter and intra pores on their surface provides a larger surface area. The larger the surface area, the higher the wettability, dissolution, and solubility of the drug. In the present work, neusilin was used as an adsorbent, which is a magnesium aluminum silicate. Neusilin has a surface area of 300 m2/g and adsorption capacity of 3.2 ml/g. Due to its amorphous nature, it restricts the conversion of amorphous to a crystalline drug. Whereas, clopidogrelbisulfate was used as a model drug as it belongs to BCS class II widely used as an antiplatelet agent or blood thinner. [3, 4]
Materials and Methods
Materials
The drug clopidogrelbisulfate was procured from Sun Pharmaceuticals Ltd., India. Polyethylene glycol – 4000, Polyethylene glycol – 6000, and Poloxamer 188 were purchased from Qualikems Fine Chem. Pvt. Ltd, India. Neusilin was gifted from Ganwal Chemicals Mumbai. Locust bean gum was purchased form Balaji chemicals, India. All chemicals were of analytical grade.
Phase solubility studies
Phase solubility studies were carried out to check the solubility of Clopidogrel bisulfate with polymers. The miscibility of Clopidogrel bisulfate with PEG 4000, PEG 6000 Poloxamer 188, and Locust bean gum was evaluated. In conical flask solutions of 2%, 4%, 6%, 8%, and 10% of each polymer was taken. An excess quantity of Clopidogrel bisulfate was added, to 20ml of each of the solutions. The solutions were shaken in an environmental shaker (at 37 °C for 24 hours). Samples were filtered with Whatman filter paper and analyzed spectrophotometrically (Shimadzu U-1800, Japan) at 220nm for the dissolved drug [5]. The stability constant was calculated from the phase solubility graph using
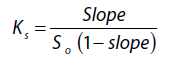
Where, Sois the solubility of Clopidogrel bisulfate in the absence of polymer. Gibb’s free energy of transfer (ΔG°) from pure distilled water to polymer solution was calculated using
Where, So/Ss is the ratio of the solubility of Clopidogrel bisulfate in the solution of polymer to that of the in absence of polymer.
32 full factorial design experiments
A three-level, two-factor 32 (3 levels, 2 factors) design was used. Low, medium, and high levels of each variable were coded as -1, 0, and +1. Nine Solid dispersion Adsorbate tablets of Clopidogrel bisulfate were prepared using 32 full factorial designs as shown in Table no:3. The range of factors must be chosen, in order to measure its effect on the response variables. The design was selected, as it provides a sufficient degree of freedom to resolve the main effects and the interaction. To find out the control factors that significantly affect response variables, Stepwise regression analysis was used. The ratio of Carrier and drug in solid dispersion (X1) and Ratio of Adsorbent to solid dispersion (X2) were selected as independent variables as shown in Table no:1, while, Q30 (Y1), angle of repose (Y2), and %DE30 (Y3) were taken as dependent variables as shown in Table no:2 [6, 7]
To calculate the response, a statical model containing interactive and polynomial terms was used as follows:
Y=b0+b1X1+b2X2+b12X1X2+b11X1X1+b22X2X2
Where, b0 is the arithmetic mean response of all trails, and b1,b2,b12,b11,b22 are the coefficient for the corresponding factor X1, X2,X1X2, X1X1, X2X2. This represents the average result of changing one factor at a time from its low to high value. Y is dependent. How the response changes when two factors are simultaneously changed, is indicated by the interaction term (X1X2). The polynomial terms (X1X1, X2X2) are included to investigate the non-linearity. The polynomial equation and all significant values were obtained by applying Design-Expert Software (7.0.0).
|
Table 1: Independent variables (And translation of coded value to the actual value) |
|||
|
Independent Variable Level |
Low (-1) |
Medium (0) |
High (+1) |
|
Ratio of Drug and Carrier in solid dispersion (X1) |
1:0.5 |
1:2 |
1:3.5 |
|
Ratio of Adsorbent to solid dispersion (X2) |
0 |
1 |
2 |
|
Table 2: Dependent variables |
|
|
Code |
Dependent variables |
|
Y1 |
Q30 (Amount of drug release at 30 min.) |
|
Y2 |
Angle of Repose |
|
Y3 |
%DE30 (30 Min.) |
Method of preparation
Solid Dispersion Adsorbate tablets of Clopidogrel bisulfate were prepared by the solvent evaporation method. PEG 4000 was dissolved in methanol. The drug was also dissolved in methanol separately. Both solutions were mixed and sonicated. Solid dispersion of Clopidogrel bisulfate and PEG 4000 were prepared in different ratios as shown in Table No.3. Neusilin was added to this mixture to obtain solid dispersion adsorbate. Then the granules were properly mixed with SSG, Spray-dried lactose, and magnesium stearate. The tablet with an average weight of 500 mg was prepared, by direct compression method using a rotary tablet machine. [8, 9]
|
Table 3: 32 full factorial design layout for Clopidogrel bisulfate containing solid dispersion adsorbate tablets (Actual value) |
||
|
Batch |
X1 (Ratio of drug and Carrier in solid dispersion) |
X2 (Ratio of Adsorbent to solid dispersion) |
|
B1 |
1:0.5 |
0 |
|
B2 |
1:0.5 |
1 |
|
B3 |
1:0.5 |
2 |
|
B4 |
1:2 |
0 |
|
B5 |
1:2 |
1 |
|
B6 |
1:2 |
2 |
|
B7 |
1:3.5 |
0 |
|
B8 |
1:3.5 |
1 |
|
B9 |
1:3.5 |
2 |
Evaluation Parameter
The dissolution was measured by USP apparatus II using 900 ml of 0.1N HCl. The medium was maintained at 37± 0.5 °C and 75 rpm. The aliquots of 5 ml were withdrawn at regular intervals. The sample was analyzed using the UV-visible spectrophotometer. [10]
It is defined as the area under the dissolution curve up to a certain time (t). It is expressed as a percentage of an area of the rectangle described by 100% dissolution at a time. The % dissolution efficiency was calculated by [11]:
Y is the drug release percentage at t time
In order to assess the reliability of the equation that describes the influence of the factors on the Q30, angle of repose, and %DE30. Additional checkpoint experiments were conducted. The % relative error between predicted values and experimental values of each response was calculated using the following equation:
To optimize all the responses with different targets, graphical optimization by the overlay plot was used. The optimized formulation was obtained by applying constraints on independent and dependent variable responses. The constraints were Q30 minimum 85% (Amount of drug release in 30 min should be more than 85%), angle of repose maximum value (Angle of repose less than 25 indicated excellent flow properties), and % DE 30 maximum value (dissolution efficiency close to 100% in 30 min). These constraints were calculated using the Design-Expert Software (Version 7.0.0) [12,13]
A differential scanning calorimetry study was carried out to evaluate the thermotropic property and thermal behavior of drug, carrier, and adsorbent
The drug, carrier, and drug-carrier mixture were prepared. The powder X-ray diffractometer was used for checking the compatibility of the drug, carrier, and drug-carrier mixture. [14]
Morphological evaluation of the Clopidogrel bisulfate and surface solid adsorbate of Clopidogrel bisulfate were carried out by scanning electron microscopy. [15]
According to ICH guidelines, the optimized formulation was kept for the accelerated stability studies (40 ± 2°C and 75 ± 5% RH), for one month in a stability chamber. The optimized formulation was placed in USP type – I amber-colored vials. Closed with rubber plugs, Sealed with aluminum caps. The samples were withdrawn after the stability period. Dissolution studies were carried out. The similarity factor (f2) was used for the evaluation of the drug release. [16]
The blood-thinning effect of Clopidogrelbisulfate on the platelet aggregation was investigated using Rat. A dose of 20 mg/kg body weight was administered orally to each rat. The control group was also treated similarly (with the sterile water at the equivalent volume relative to body weight). Cotton was soaked with chloroform solution and put close to the animal nose in order to keep the animal anesthetized. The tail of the rat was cut (0.5 cm distal tip of the tail) and placed in a pre-warmed phosphate buffer saline solution. The time of onset of bleeding and collection of blood volume was recorded. [17]
Result and Discussion
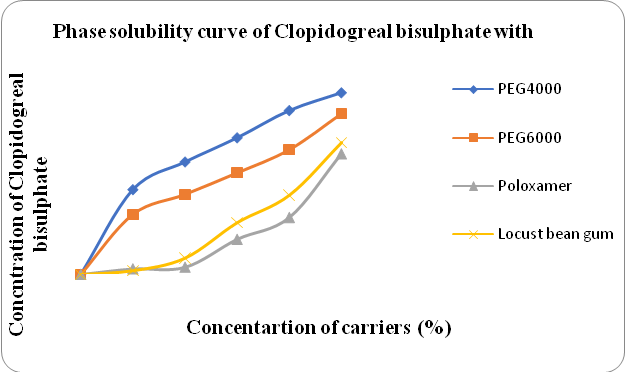
Figure 1: Phase solubility study of Clopidogrel bisulfate with different polymers
Figure No:1 shows the phase solubility of Clopidogrel bisulfate in different Carriers. The R2 value was found highest in PEG 4000.
|
Table 4: Gibb’s free energy transfer for the solubilization process of Clopidogrel bisulfate in different Carriers |
||||
|
Carrier Concentration (%) |
Gibb’s energy transfer KJ/mol at 37ºC |
|||
|
PEG 4000 |
PEG 6000 |
Poloxamer 188 |
Locust bean gum |
|
|
2 |
-1.014 |
-0.58 |
-0.56 |
-0.535 |
|
4 |
-1.06 |
-0.74 |
-0.65 |
-0.429 |
|
6 |
-1.10 |
-1.0 |
-0.73 |
-0.658 |
|
8 |
-1.15 |
-1.088 |
-0.76 |
-0.956 |
|
10 |
-1.16 |
-1.12 |
-0.80 |
-0.914 |
|
Table 5: Solubility obtained at 37oC from different Carriers |
|||
|
Carrier |
Slope |
Stability constant |
r2 |
|
PEG 4000 |
0.5722 |
8.91 |
0.9582 |
|
PEG 6000 |
0.4854 |
6.28 |
0.9369 |
|
Poloxamer 188 |
0.4169 |
4.76 |
0.9132 |
|
Locust bean gum |
0.3556 |
3.67 |
0.8283 |
The solubility of Clopidogrel bisulfate in different carriers was evaluated from phase solubility studies and it was found to be 0.15 mg/ml in water. The solubility increased with increasing the concentration of polymer. The r2 value of PEG 4000 was 0.95 (Table no:5), which was the highest compared to other polymers. The negative sign of Gibbs free energy showed the solubilization process of Clopidogrel bisulfate. PEG 4000 was used as a carrier for the preparation of solid dispersion as shown in Table no:4. [18]
Figure No 2 shows the drug release of Clopidogrel bisulfate containing different ratios of PEG 4000 prepared using the solvent evaporation method. In both methods, different ratios of polymer, i.e. 1:1, 1:2, 1:4, and 1:6 were used. The drug release of the S2 batch was the highest. Compared to the hot-melt method, the solvent evaporation method gave better drug release. The drug release was also increased in surface solid dispersion as compared to plain API and solid dispersion. Also, when the ratio of drug: polymer increased from 1:1 to 1:2, there was an improvement in drug release. Compared to the 1:2 ratio, when 1:6 ratio was used, the drug release decreased.
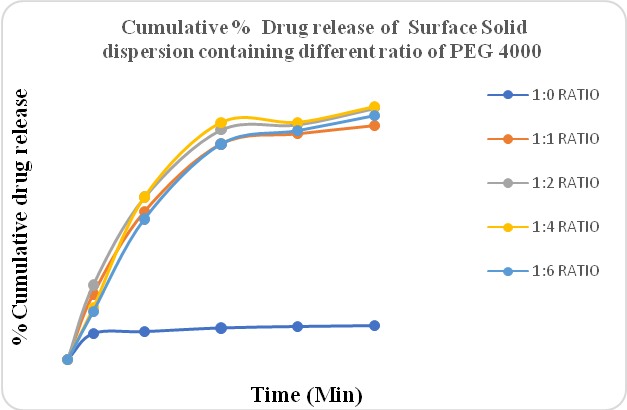
Figure 2: %CPR of Surface solid dispersion containing different ratios of PEG 4000 (Solvent evaporation method)
Figure No.3 shows the drug release of Clopidogrel bisulfate containing different ratios of PEG 6000 prepared by the solvent evaporation method. Different ratios of polymer, 1:1, 1:2, 1:4, and 1:6 were used. The drug release of the solvent evaporation method was better than the hot-melt method. The PEG 4000 and PEG 6000 gave better drug release compared to Poloxomer 188.
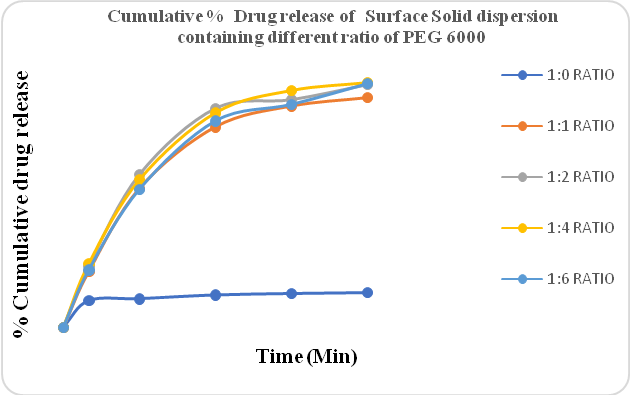
Figure 3: % CPR of Surface solid dispersion containing different ratios of PEG 6000 (Solvent evaporation method)
Figure No.4 shows the drug release of Clopidogrel bisulfate containing different ratios of Poloxomer 188 prepared by solvent evaporation method. In both methods, different ratios of polymer 1:1, 1:2, 1:4, and 1:6 were used. The PEG 4000 and PEG 6000 gave better drug release than Locust bean gum. The drug release of Poloxomer 188 was less than Locust bean gum.
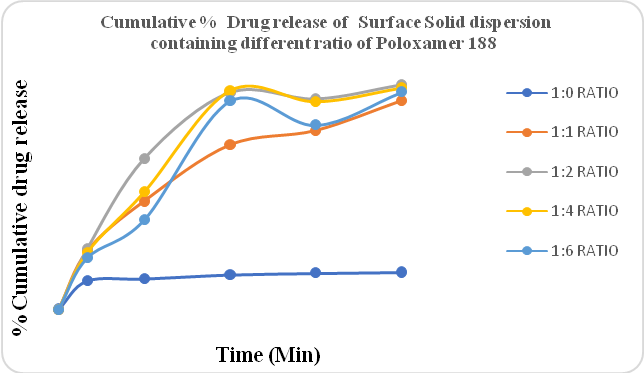
Figure 4: %CPR of Surface solid dispersion containing different ratios of Poloxamer 188 (Solvent evaporation method)
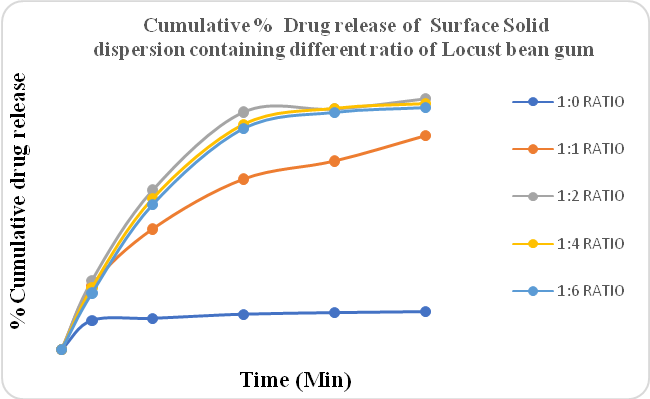
Figure 5: %CPR of Surface solid dispersion containing different ratios of Locust bean gum (Solvent evaporation method)
Figure No.5 indicated the drug release of Clopidogrel bisulfate containing different ratios of Locust bean gum prepared by the solvent evaporation method. As per USP, the amount of drug release for conventional formulations of Clopidogrel bisulfate in 30 min should be more than 85%. The cumulative % drug release of Locust bean gum, poloxamer 188, and also PEG 6000 was less than 85%. PEG 4000 containing Surface solid dispersion gave a drug release of more than 85%. The amount of drug release increased compared to solid dispersion in surface solid dispersion. The drug:polymer ratio of 1:2 gave the highest amount of drug release in PEG 4000. Also from the result, it was concluded that the amount of drug release in the solvent evaporation method was better than the hot melt method.
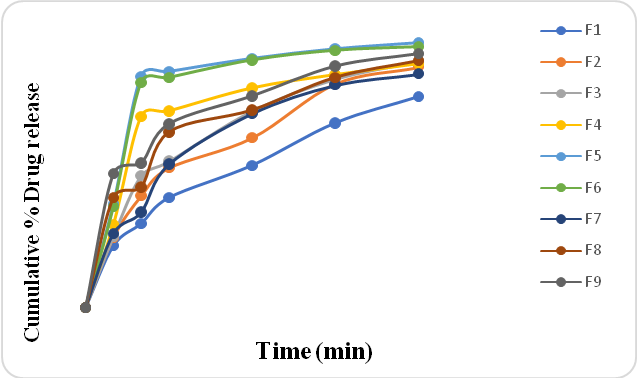
Figure 6: % CPR of Clopidogrel bisulfate containing surface solid dispersion tablets
Figure No.6Shows the drug release of solid dispersion adsorbate tablets containing the drug. There was an increment in drug release withincreasing polymer. As per USP, the amount of drug release in 30 min for the conventional formulation is 85%. Formulation F5 and F6 and F9 showed a drug release of more than 85%.
Result of ANOVA study for Q30
The results indicated that the model is significant. The model F-value of 32.73 implied that the model is significant. There was only a 0.81% chance that a “model F-value” large could occur due to noise. Values of Prob>F less than 0.0500 indicate model terms are significant. In this case, A, B, and A2 are significant model terms. Values of greater than 0.1000 indicate the model terms are not significant.
Polynomial equation for Q30(Y1)
Y = + 86.40 + 5.41A + 6.05B + 3.31AB - 18.71A2 - 1.53B2 (Coded value)
Y = + 33.91 + 39.07A + 13.53B + 2.20AB – 8.31A2 – 1.53 B2 (Actual value)
As shown in Figure 7, The coefficient values indicate that, as the Ratio of Carrier and Drug in solid dispersion increases, the amount of drug release at 30 min increases. Due to PEG 4000, the dissolution rate of Clopidogeral bisulfate increases. When the PEG comes in contact with the dissolution medium, it becomes hydrated. This causes the solubilization of drug particles and releasing the drug. The positive sign indicates that the drug release increased with increasing concentration of the carrier.
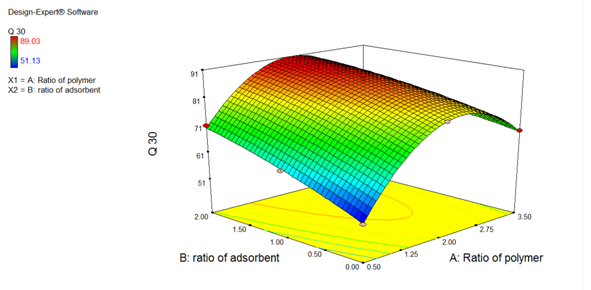
Figure 7: Response Surface Plot (Effect of formulation variable on Q30)
Results of ANOVA study (Angle of repose)
As shown in Figure 8, The model F-value of 10.94 implies that the model is significant. There is only a 3.84 % chance that a “Model F-value” large could occur due to noise. “prob > F” values of less than 0.05000 indicate that the model terms are significant. In this case, B is a significant model term. Values of greater than 0.1000 indicate that the model terms are not significant.
Polynomial equation for Angle of repose
Y = + 25.18 – 1.83A – 9.63B + 1.84AB + 3.13 A2 + 5.93B2 (Coded value)
Y = +51.18 - 8.00 A – 23.93B + 1.22 AB + 1.38 A2 + 5.92B2 (Actual value)
The Bhas a higher value than A. The value of less than 25 indicates excellent flow properties. The negative value of B indicates that the solid dispersion mass was not adsorbed on the surface of Neusilin. Thus, this increases the flow property. The higher value of the angle of repose shows poor flow property because the solid dispersion mass obtained was sticky. The angle of repose decreased with increasing the adsorbate ratio.
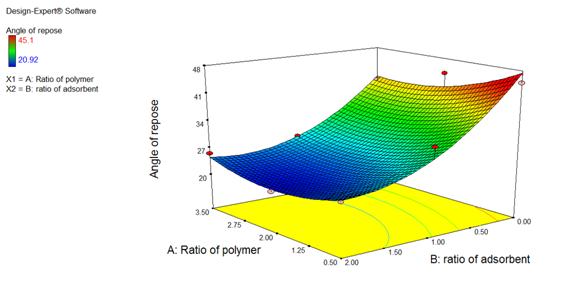
Figure 8: Response Surface plot of Angle of repose)
Result of ANOVA study (%DE30)
The model F – values of 17.31 implies the model is significant. There is only a 2.02% chance that a “ Model F-value” large could occur due to noise. The “Prob > F” values of less than 0.0500 indicate that the model terms are significant. In this case, A, A2 were significant model terms.
Polynomial equation for %DE30
Y = + 92.81 + 4.71A + 3.05B – 2.77AB – 15.56A2 + 0.63B2 (Coded value)
Y = + 52.76 + 32.64 A+ 5.47B – 1.84AB – 6.91A2 + 0.63B2 (Actual value)
As shown in Figure 8, The result of the ANOVA study showed that dissolution efficiency increased with increasing the amount of Carrier and Adsorbent. The Carrier has more effect on the dissolution than Adsorbent. The positive sign indicates that the dissolution was improved with increasing the amount of Carrier and Adsorbate.
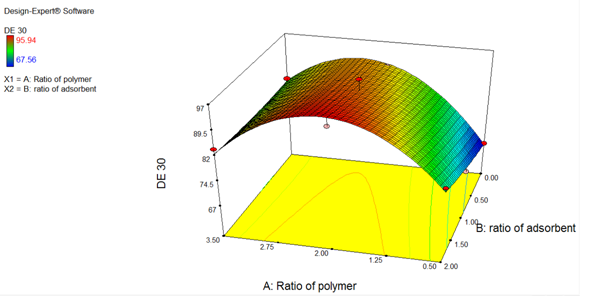
Figure 9: Response surface plot of (%DE30)
|
Table 6: Statistical results obtained from ANOVA study |
||||
|
Response |
Adjusted R2 |
Predicted R2 |
Adequate Precision |
C.V (%) |
|
% DE30 |
0.9107 |
0.7354 |
12.508 |
3.44 |
|
Angle of repose |
0.7387 |
0.6230 |
8.200 |
1.51 |
|
Q30 (Amount of drug release at 30 min) |
0.9520 |
0.8439 |
18.314 |
3.63 |
In Table 6, the response of %DE30, Angle of repose and Q30( Amount of drug release at 30 min) have Coefficient of variance value of 3.44, 1.51 and 3.63 respectively , which shows that all results are in acceptable range.
Model validation
To validate the model, Checkpoint batches were prepared and evaluated as shown in Table no:7. The condition was the same as the outline for the other batches. The results were found within acceptable criteria.
|
Table 7: Comparison between the observed and predicted values of the checkpoint batch of Clopidogrel bisulfate |
||||||
|
Sr. No. |
X1 |
X2 |
Parameters |
Predicated |
Observed |
Bias (%) |
|
1 |
1:1.29 |
1.94 |
Q30% |
85.51 |
83.20 |
2.70 |
|
Angle of repose |
22.10 |
21.47 |
2.85 |
|||
|
% DE30 |
91.80 |
89.63 |
2.17 |
|||
|
2 |
1:1.07 |
0.54 |
Q30% |
71.83 |
70.59 |
1.72 |
|
Angle of repose |
33.71 |
32.34 |
4.06 |
|||
|
%DE30 |
81.87 |
80.43 |
1.75 |
|||
Selection of optimization batch
The overlay plot was used to obtain all the responses with different targets (byusing a multicriteria decision approach). The constraints were %DE30 (dissolution efficiency of 95%), Q30 minimum 85% (the amount of drug release in 30 min for Clopidogrel bisulfate should be more than 85% (AS per USP) and for Angle of repose maximum value 25 (value of less than 25 shows excellent flow property). By applying constraints on dependent variable responses and independent variables the optimized formulation was obtained. The variables were calculated using Design-Expert (version 7.0.0). The overlay plot is shown in Fig No.9.1
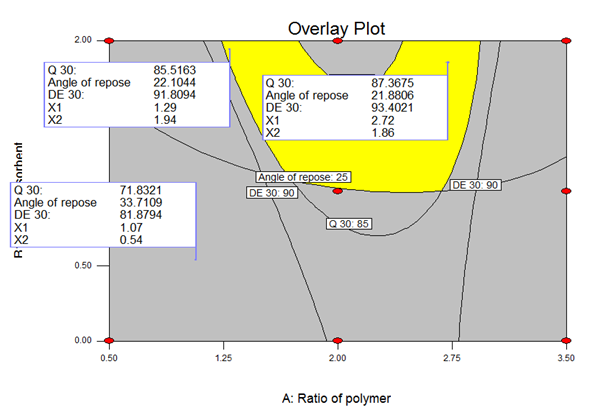
Figure 9.1 Overlay plot of Optimized batch of Clopidogreal bisulphate containing Surface Solid dispersion tablets
In the overlay plot, the yellow portion shows the design space for the said target values.
|
Table 8: Observed and experimental values of the optimized batch of Clopidogrel bisulfate containing Surface solid dispersion tablets |
|||||
|
X1 |
X2 |
Parameters |
Predicted |
Obsereved |
% Bias |
|
1:2.72 |
1.86 |
% DE30 |
93.40 |
89.64 |
4.02 |
|
Angle of repose |
21.88 |
21.37 |
2.33 |
||
|
Q30 |
87.36 |
85.69 |
1.91 |
||
Table no.8 shows observed and experimental values of the optimized batch of Clopidogrel bisulfate.
Overlay plot of Optimized batch of Clopidogrel bisulfate containing Surface Solid dispersion tablets
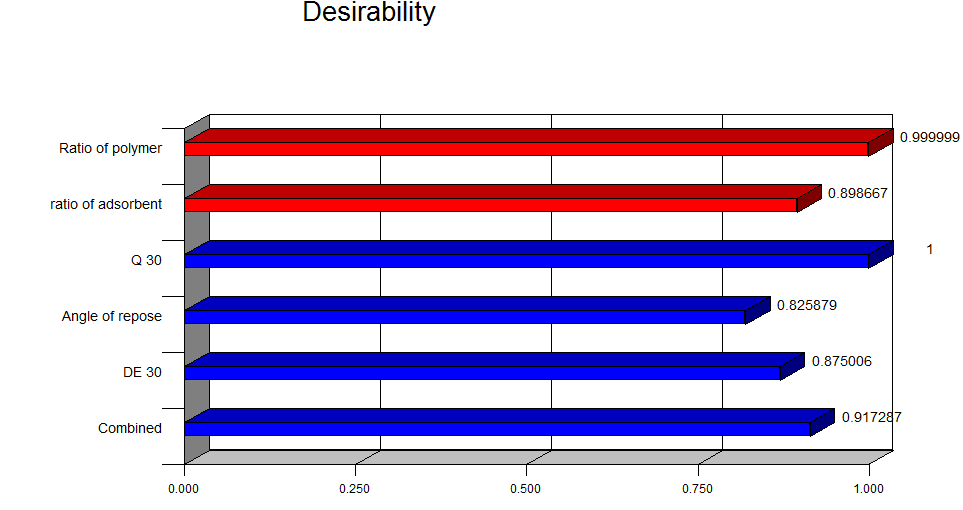
Figure 10: Desirability plot of Clopidogrel bisulfate containing Surface solid dispersion tablets
Figure no.10 shows the desirability plot of Clopidogrel bisulfate containing surface solid dispersion tablets.

Figure 11: Scanning Electron Microscopy of Solid dispersion Adsorbate
Figure no.11 shows the surface morphology of solid dispersion adsorbate of Clopidogrel bisulfate. The irregular shaped crystalline agglomerate of Clopidogrel bisulfate was found. Neusilin having many inter and intra particular pores on its surface provides a much larger surface area. The surface solid dispersion showed no agglomeration and free-flowing property.[19]
|
Table 9: Result of Gas Chromatography |
|
|
Sample Name |
Methanol (PPM) |
|
Surface Solid Dispersion |
2.33 |
As shown in Table no.9, the amount of methanol present in Surface solid dispersion was 2.33 PPM, which os below the limit (proved by Gas chromatography). So, methanol was used as a solvent for the preparation of Surface Solid dispersion.[20]
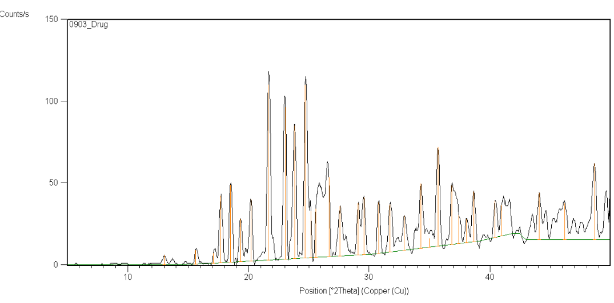
Figure12: X-ray diffraction study of Clopidogrel bisulfate
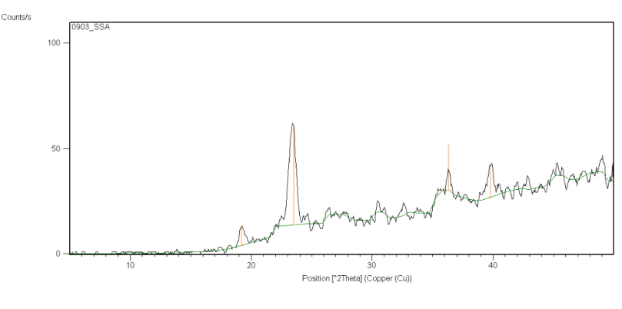
Figure 13: X-ray diffraction study of Clopidogrel bisulfate containing Surface Solid Dispersion
The diffraction pattern of Clopidogrel bisulfate exhibited an intensity peak at, 2θ values of 13.03º, 15.62º, 17.08º, 17.69º, 18.48º, 20.9º, 25.16º, 25.69º as shown in Figure no.12. This indicates the crystalline nature of Clopidogrel bisulfate. In Surface solid dispersion, it did not show the peak, which was present in Clopidogrel bisulfate. So, enhancement of the dissolution of the drug was attributed to decreased crystallinity of the drug as shown in Figure no.13.
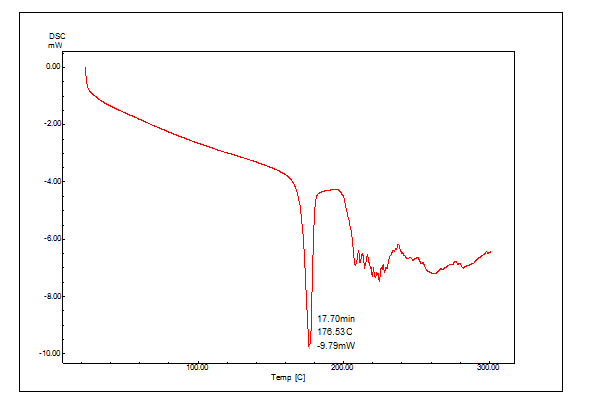
Figure 14: DSC thermogram of Clopidogrel bisulfate
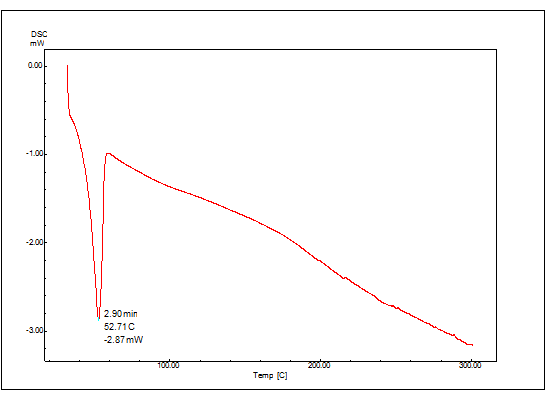
Figure 15: DSC thermogram of Clopidogrel bisulfate containing Surface Solid Dispersion
The DSC thermogram of Clopidogrel bisulfate and surface solid dispersion is shwon in Figures no:14 and 15, respectively. Clopidogrel bisulfate shows a sharp peak at 176.63º C corresponding to its melting point (176 -178º C). In figure no:15 the complete disappearance of the drug melting peak in surface solid dispersion was found because the drug solubilized completely in carrier PEG 4000.
|
Table 10: Comparison of prepared tablets of Clopidogrel bisulfate (Before and After Stability Studies) |
||
|
Parameters |
Initial |
After 30 days |
|
Room Temperature |
40° C, 75% RH |
|
|
%DE 30 |
86.21±0.25 |
84.27±0.21 |
|
Q30 |
90.31±0.39 |
89.31±0.19 |
The results in Table no.10 show that %DE30 and Q30 did not change after 30 days of storage.
The inhibition of platelet aggregation was measured using the bleeding time.
|
Table 11: Platelet aggregation inhibition study |
||
|
Sr.No. |
Bleedingtime |
Blood volume |
|
Control group(Without treatment) |
90 sec |
0.9 ml |
|
Test group – (Clopidogrel bisulfate) |
3 min |
0.12 ml |
|
Test group – (solid dispersion containing Clopidogrel bisulfate) |
7 min |
1.3 ml |
|
Test group – (surface solid dispersion containing Clopidogrel bisulfate) |
7.5 min |
1.5 ml |
Table no.11 shows the bleeding time of rats. The bleeding time of the control group was 90 sec. while it was 3 min in the rats treated with Clopidogrel bisulfate (plain drug). Moreover, it increased in the case of solid and surface solid dispersions. The collected blood volume increased when the rats were treated with Clopidogrel bisulfate, solid dispersion containing Clopidogrel bisulfate and also surface solid dispersion. [17]
Conclusion
From this study, it was concluded that the solubility and dissolution properties of Clopidogrel bisulfate were improved by using the solid dispersion Adsorbate technique. By response surface plot analysis and polynomial equation, the effect of independent variables on dependent variables (dissolution variables) such as %DE30, Angle of repose, and amount of drug release in 30 min was analyzed. The factorial batches were evaluated for pre- and post-compression parameters. From the platelet aggregation inhibition study, the effectiveness of Clopidogrel bisulfate containing surface solid dispersion was confirmed. So, it can be concluded that the Solid Dispersion Adsorbate method is one of the alternative methods for improving solubility and dissolution of poorly water-soluble Clopidogrel bisulfate.
Conflict of Interest
There is no conflict of interest for this research work.
References
